Healthcare, Medical & Life Sciences
Tecman has been supporting the medical industry for over 30 years, converting FDA approved materials…
Securing supply continuity, cost, and quality of a medical device
"The customer approached us to take over manufacture of a medical component to ensure no break in supply and no price increase. This presented a significant challenge, however we committed to support the customer and they were delighted with what we were able to achieve. Our efforts have been rewarded with a long-term supply agreement and we’re now looking at optimising the component further to boost the customer's competitive edge."
Alan Bryan, Key Account Manager
A UK medical device manufacturer faced a critical supply chain disruption when their long-standing material supplier announced they would no longer convert their raw material into a critical die-cut adhesive component. The affected product was a Class I medical device that had a loyal and growing user base making uninterrupted supply essential.
The manufacturer urgently needed a proven partner to step into the supply chain. So, they approached Tecman with a clear set of requirements:
To deliver a seamless and disruption free supplier switchover, we initiated a highly collaborative process with both the customer and the material supplier. This joint approach enabled us to fully understand the component, materials, packaging and supply conditions, and all other associated requirements including legal and regulatory.
To achieve cost neutrality with design and materials fixed, we focused on maximising manufacturing and material efficiency. The first step in achieving this was visiting the previous supplier and collaborating on multiple test runs to identify the ideal material supply format.
Secondly, the Tecman team identified an opportunity to improve the yield from the material, effectively gaining the customer more parts from the same material. The focus then lay on the manufacturing process, and our larger machines enabled a wider web, producing more parts in the same amount of time.
Following this, we invested in some end-of-line automation to further improve the run speeds, but also reduce the manual resources needed. These developments significantly reduced material waste and maximised efficiency, successfully offsetting the transition costs.
To support long-term, reliable production of the component, we made targeted enhancements to our clean manufacturing environment. This ensured that all quality, cleanliness, and regulatory requirements would be consistently met.
Tecman successfully took over production in just three months, maintaining exact quality and cost parity—an impressive feat given the previous supplier was the material manufacturer. By improving yields and investing in automation, we offset all transition costs and secured an uninterrupted supply chain.
The customer was delighted with the switchover and has since awarded Tecman a long-term supply agreement. We have now moved into the next phase: implementing a strategic roadmap of further cost-savings to help our customer achieve their goal of becoming a global market leader.

Faced with a sudden, critical supply chain disruption and demanding constraints regarding cost and quality, the customer needed a proven solution-driven partner.
They chose Tecman because of our agility and collaborative approach, which enabled a seamless, disruption-free entry into the supply chain. Our technical expertise was key; we developed innovative manufacturing processes, featuring the novel material-guiding system, to significantly reduce waste and achieve cost neutrality while maintaining component quality. Furthermore, our targeted investment in our cleanroom facilities demonstrated our commitment to the regulatory requirements.
We can support our customers through every stage, from material selection and design phases to choosing the right manufacturing processes. This ensures the delivery of an optimised, cost-effective solution designed to effectively overcome challenges and deliver tangible benefits.
If your application could benefit from cost savings, process improvements, material enhancements, and product advancements, reach out to our experts to discover how we can support your application.

Tecman has been supporting the medical industry for over 30 years, converting FDA approved materials…
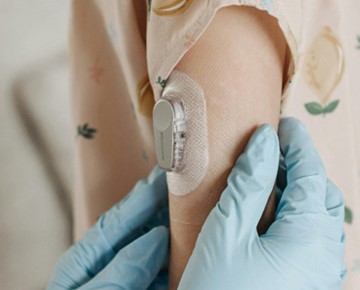
Custom die-cut stick-to-skin components for wearable medical devices.

Tecman has a track record of working with diagnostic manufacturers to effectively develop engineered…

The medical diagnostics industry faces the critical task of enhancing sustainability. Can the lesson…

We improved LSE bonding performance and production efficiency for a SME customer through developing …

We developed a custom die-cut protection film component that eliminated damage during transportation…

Design & manufacture of a custom die-cut adhesive gasket to bond a plastic component to a vehicl…
Our proven track record underlines our commitment to delivering value-added products with consistent…